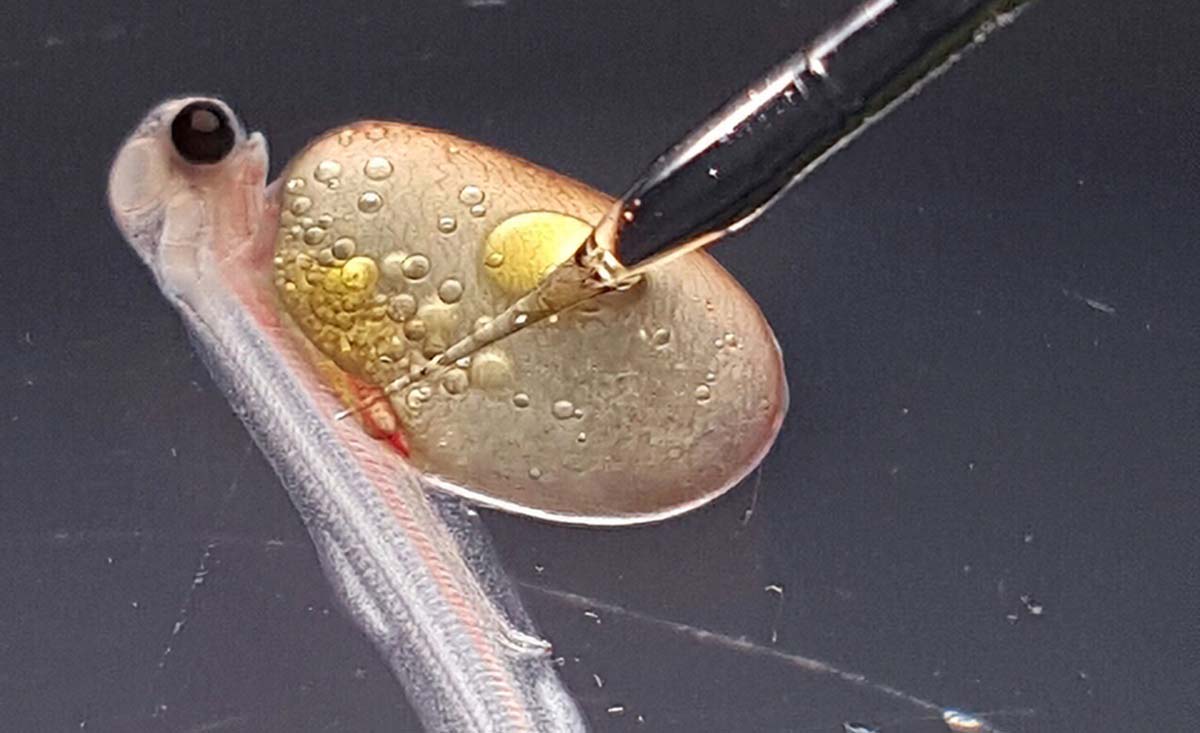
Researchers are learning how to produce fish with surrogates from another species. Here, a researcher microinjects germ cells into the peritoneal cavity of a newly hatched rainbow trout. Photo courtesy of Tokyo University of Marine Science and Technology
Surrogacy: Scientists can now borrow the bodies of one fish species to produce another—whether they should, though, is an open question.

Chloe Williams is a science journalist based in Edmonton, Alberta, covering the environment and health. Her work has been featured in Popular Science, Audubon, and Yale Environment 360, among other publications. When she’s not hunched over behind her desk, you’ll likely find her chasing adventures in the outdoors.
By Chloe Williams / Hakai Magazine / September 28, 2021
This article is also available in audio format. Listen now, download, or subscribe to “Hakai Magazine Audio Edition” through your favorite podcast app.
NOTE: The following is an excerpt
For the past 20 years, Yoshizaki, a professor of marine biosciences at Tokyo University of Marine Science and Technology in Japan, has been developing a way to borrow the body of one species to produce the sperm and eggs of another. Just as a doctor might borrow the uterus of a surrogate mother to nurture a human embryo, Yoshizaki borrows the gonads of a surrogate fish to cultivate eggs and sperm, or milt.
Chinese rosy bitterling are one of his latest experiments. The species, invasive in Japan, outcompetes native bitterling. When the invasive bitterling spawn in Yoshizaki’s lab, they’ll produce the sperm and eggs of the Tokyo bitterling, an endangered species considered a “national monument,” a federal designation that gives it special protection. “It’s an ironic situation,” he says. The invasive bitterling will reproduce the very species they are driving to extinction in the wild.
These fish won’t be the first to produce gametes—eggs and sperm—that aren’t their own. In 2004, Yoshizaki managed to get salmon to spawn trout. He and his colleagues have since used surrogacy to successfully breed several other species, including pufferfish and Japanese yellowtail, and researchers at other institutions have been exploring its use in sturgeon, carp, and trout.
A future that uses surrogates to aid in aquaculture or conservation could have major implications
Surrogates may allow fish farmers to breed large species in smaller bodies, cutting the cost of producing fish for human consumption. Conservation biologists could use common species to spawn endangered ones. Beyond breeding fish in unexpected ways, the cells researchers implant in surrogate parents can be cryogenically stored, which means scientists can bank the genetic information of species and use it to spawn more of those same fish in the future—an elusive goal they’ve been pursuing for decades. Surrogates could help save species.

Will the future be trouts galore delivered by carp? Illustration by award winning water colorist Thom Glace.
Whether scientists should use them to do that, however, is less clear
The idea of freezing cells to save them goes back centuries, but the first major successes in cryopreservation date back to the mid-1900s. In 1949, researchers in London froze fowl semen, and after thawing, the sperm kept right on swimming. Soon after, a cow gave birth to the first calf conceived from frozen-and-thawed sperm. Researchers named it Frosty.
To make a surrogate, Yoshizaki collects early germ cells from the gonads of a donor fish and transplants them into another fish. There, the immature cells develop into sperm or eggs. By pairing male and female surrogates, researchers can breed new fish that match the donor species. And by combining surrogacy with cryopreservation, scientists can avoid freezing eggs or embryos. Instead, they can freeze early germ cells and thaw them before transplant. The cells’ small size and simple structure makes them easy to cryopreserve. Once, Yoshizaki and his colleagues stuck a whole trout in a deep freezer without any chemical protectants. Two years later, when the team pulled the fish from the freezer, they were surprised to discover that they could still harvest the cells they needed to produce the frozen trout’s eggs and sperm.
Finding a surrogate capable of incubating a foreign species’ cells, however, is less precise. The surrogate and the donor should belong to the same genus or family—if researchers stray too far on the evolutionary tree, the cells won’t develop into eggs or sperm. Ideally, a surrogate is also sterile, so that when it reaches maturity it produces only the donor’s gametes. If the procedure is successful, the donor’s cells migrate inside the surrogate’s body, guided by chemical cues, to its ovaries or testes, where the cells multiply and mature. Regardless of the donor’s sex, those cells can turn into eggs or sperm. Cells transplanted into a male fish develop into sperm; in a female, they turn into eggs.
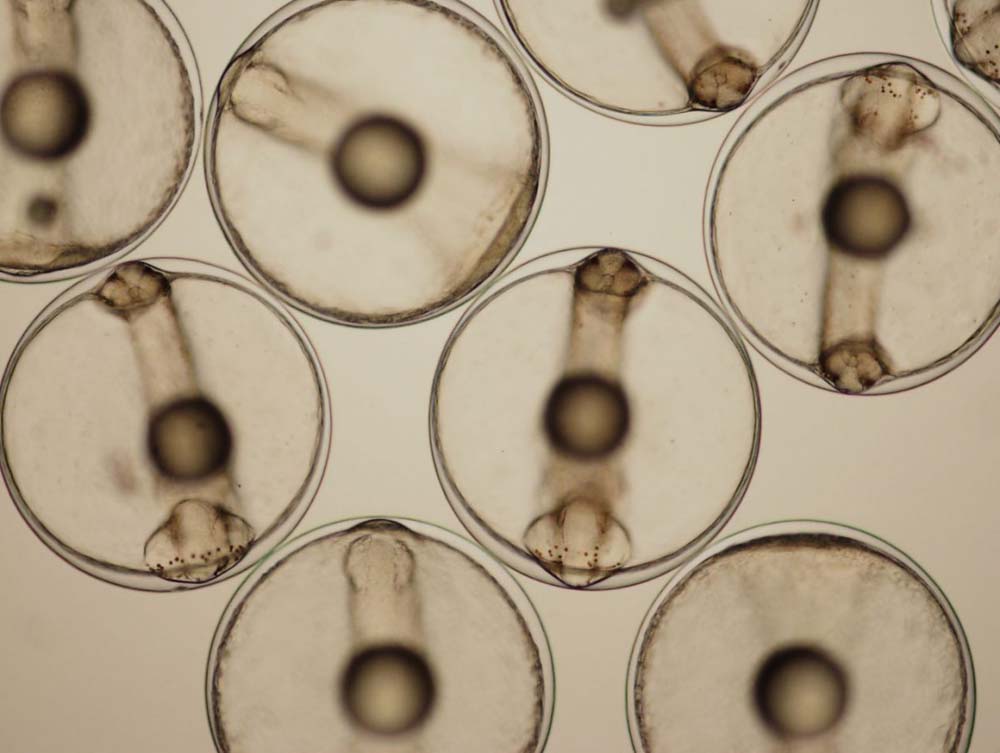
Yoshizaki and his colleagues edited the genomes of these Nibe croaker embryos to sterilize them so that they can be used as surrogate parents. Photo courtesy of Tokyo University of Marine Science and Technology
Since 2004, when Yoshizaki first spawned trout from salmon, the concept of cross-species parenthood has steadily gained momentum. In the Czech Republic, scientists are using surrogates to help save sturgeon, the most critically endangered group of species on the planet, according to the International Union for Conservation of Nature (IUCN). Some sturgeon take more than 20 years to reach sexual maturity, which makes restoring their populations challenging. For some species, surrogates may be the last hope for recovery, says Martin Pšenička, a professor of fisheries and protection of waters at the University of South Bohemia in the Czech Republic, who is leading the work.
So far, researchers have only established surrogates for a handful of species, but that hasn’t stopped them from cryopreserving samples with the goal of one day finding the right surrogate parents. In Japan, Yoshizaki has frozen cells from some of the country’s endangered fish species, including three types of bitterling, a rare species of stickleback found in a single creek, and kunimasu salmon, a species previously thought to be extinct.
If fish farmers get the go-ahead to use surrogates more widely, it could make aquaculture more efficient. Typically, fish used for breeding are kept alive in tanks or pens, a considerable expense for large, slow-growing species. “By using surrogates, we can save a lot of space and labor,” Yoshizaki says. Surrogates could also help take pressure off wild fish stocks. To bring attention to the technique’s potential, Yoshizaki is chasing a flashy example: he wants to use mackerel to spawn Pacific bluefin tuna. Although the two species have similar egg sizes, tuna broodstock individuals can reach upward of 100 kilograms, while mackerel average only 300 grams, making them much cheaper to maintain.
Is an endless supply of threatened with extinction gamefish just around the corner? Envato image.
Last November, Yoshizaki made the latest addition to his own bank of frozen genetic material: rare Japanese char. He captured some of the fish in a stream north of Tokyo and carried them back to the city in a tank in the trunk of his car. Back in the lab, he dissected the char, placed their gonads in vials, and submerged them in liquid nitrogen. Now, the samples sit in a metal vat alongside frozen cells from a handful of other species. His collection is only the start of a monumental and critical task—preserving the genetic diversity of fish. One in 16 marine fish species is currently listed as threatened by the IUCN, and in 2020 alone, 16 species of freshwater fishes went extinct globally. “We need some backup,” Yoshizaki says. While he waits for the world to catch up to the idea of fish being born from surrogate parents, he plans to keep saving what he can.



